How Manufacturers Fix Broken Quality Management
Why Quality Management Is Broken (And What Needs to Change)

The audit is coming. The answers aren't.
It starts the same way every time.
An audit gets scheduled. A customer asks for documentation. A deviation triggers an investigation. Suddenly, the organization shifts into reactive mode.
Quality leaders begin searching.
They open shared drives, dig through folder structures, and try to confirm whether the latest SOP is actually the latest. They check spreadsheets tracking CAPAs. They search emails for approvals. They message colleagues to confirm whether training was completed after the last change.
At the same time, operations teams are asking questions of their own: Is this the right version of the work instruction? Has this process already been updated? Who owns this corrective action?
Everyone is working hard, but no one is working from the same place.
This is what quality management looks like in many manufacturing organizations today. Not because teams don't care about quality, but because the systems they rely on were never designed for how quality actually works.
The real problem isn't compliance. It's fragmentation.
Most organizations don't have a single "quality problem." They have a fragmentation problem.
Quality processes are spread across shared drives for documents, spreadsheets for CAPA and audit tracking, email chains for approvals, ERP or MES systems for operational data, and separate tools for training records. Each of these systems plays a role, but none of them connect the full picture.
Documents exist without context, workflows happen without visibility, and evidence is created but not captured in a structured way.
So when someone asks a simple question
"Are we audit-ready right now?"
the answer requires hours, sometimes days, of effort.
This fragmentation creates what many teams experience daily but rarely name directly: operational friction.
What operational friction actually looks like
Operational friction isn't just inconvenience, it has real consequences across the business.
Time is lost searching for documents instead of improving processes. Approvals are delayed, slowing production or product release. CAPAs take longer to close because information is incomplete or scattered.
And it becomes most painful during audits.
Audit preparation turns into a scramble. Teams rush to gather evidence that should already exist. Documents are checked and rechecked, training records are manually verified, and gaps are discovered late.
The risk isn't just inefficiency, it's exposure in the form of missing documentation, outdated procedures, and incomplete traceability.
These aren't edge cases. They're symptoms of a deeper issue: quality systems that manage files, but not context.
Why traditional QMS approaches fall short
Many organizations have tried to solve these problems by implementing a Quality Management System. But even then, the challenges persist.
This is because traditional QMS platforms often replicate the same limitations in a different interface. Documents are still treated as static files, workflows still rely on manual processes, and adoption often struggles outside the quality team. Most importantly, they fail to connect all the moving parts of quality into a single, structured system.
The result is a paradox. Organizations have a QMS, but still rely on spreadsheets, chase approvals in email, and prepare manually for audits. The system exists. but the work happens elsewhere.
The missing piece: context
At the core of these challenges is something subtle but critical: context.
A document on its own doesn't tell you which product it applies to, which supplier it affects, whether training has been completed, which CAPAs are related, or whether it's the currently approved version across all sites.
That knowledge lives in people's heads, or in disconnected systems.
Without context, quality becomes guesswork. Teams spend time connecting dots instead of executing work, decisions are made with incomplete information, and the organization remains reactive instead of proactive.
A shift is happening in how quality is managed
Forward-looking manufacturers are starting to recognize that the issue isn't just tools, it's the model.
Instead of organizing quality around folders, systems, or isolated workflows, they are shifting toward organizing it around context. This means connecting documents to products, processes, suppliers, and sites; structuring workflows so actions, approvals, and evidence are linked; capturing relationships between audits, CAPAs, training, and documentation; and making information discoverable based on what it is, not where it's stored.
This is the foundation of a context-first approach to quality management, and it changes everything.
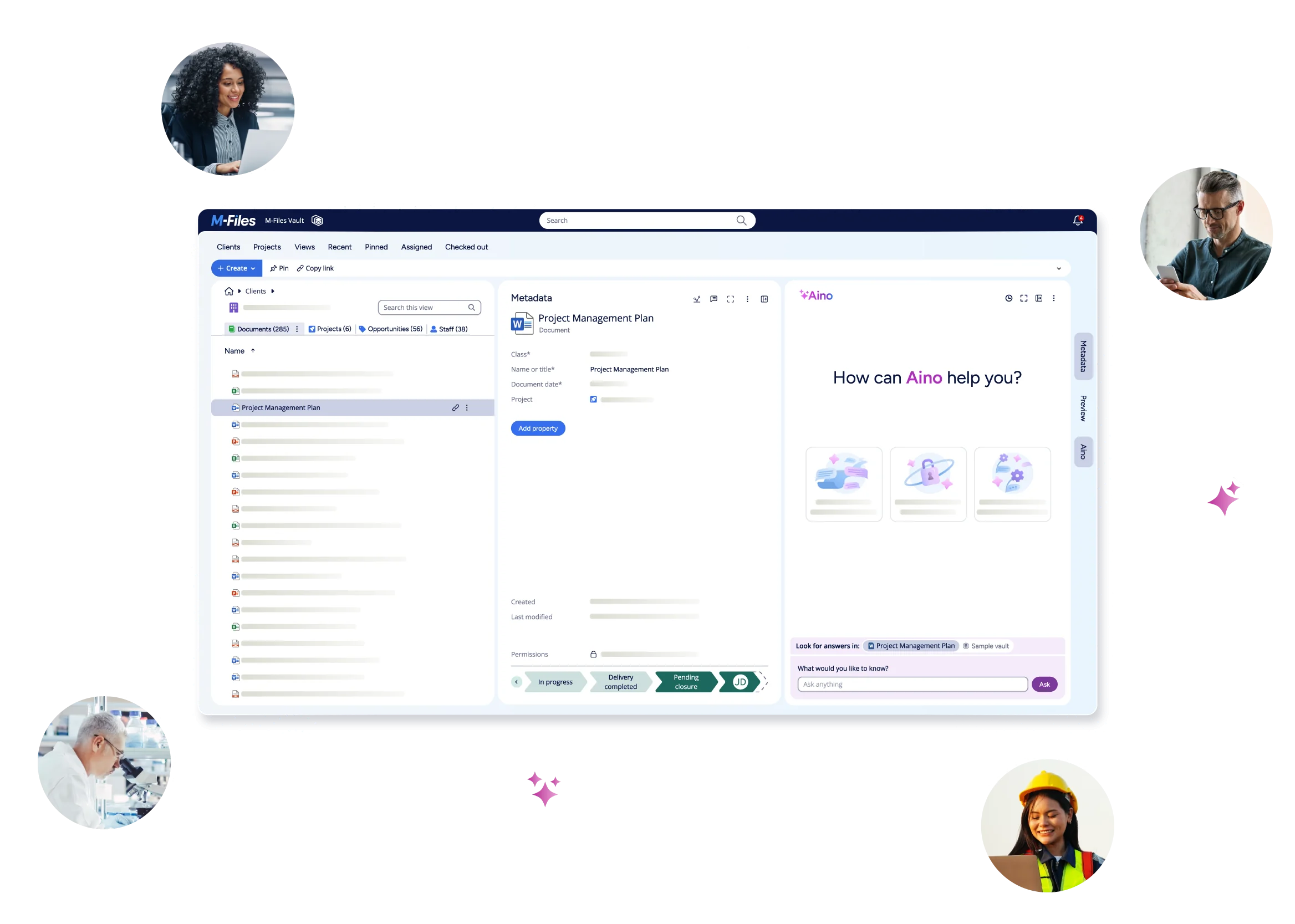
Introducing a different approach: M-Files for Quality
M-Files for Quality is built around this exact shift.
Instead of asking teams to manage files across multiple systems, it organizes quality work around connected, governed context. Documents, CAPAs, audits, training records, and supplier information are not stored in isolation - they are linked, structured, and managed as part of a unified system.
This means that when a document changes, training requirements can trigger automatically. When an audit is conducted, evidence is already connected. When a CAPA is initiated, it is tied to the originating issue, related documents, and responsible teams.
The result is not just better organization; it's a fundamentally different way of working.
From reactive firefighting to proactive quality execution
When quality is managed through context instead of disconnected systems, something important happens.
Work becomes visible, ownership becomes clear, and evidence is created as part of the process, not collected after the fact. Audit readiness becomes a continuous state rather than an event.
Instead of scrambling to answer questions, teams can respond instantly with confidence. Instead of chasing information, they can focus on improving processes. Instead of quality being seen as a bottleneck, it becomes a driver of operational performance.
The bottom line
Manufacturers don't need more tools, and they don't need another system layered on top of existing complexity.
What they need is a way to bring everything together - documents, workflows, and evidence - into a single, structured, and connected environment.
They need quality context.
Because when quality is built on context, everything else follows: better decisions, faster execution, stronger compliance, and far less scrambling when the next audit comes.
See how M-Files for Quality helps you eliminate operational friction and achieve continuous audit readiness.